THE COLLAGEN-ELASTIN MATRIX IN SKIN AGING
Laura Masini, JetPeel by TavTech Global Educator
ABSTRACT
The collagen–elastin matrix constitutes the structural and functional framework of the dermal extracellular matrix and is essential for skin stability and elasticity. This network integrates collagen fibers, elastic fibers, and a hydrated extracellular environment, which together regulate force distribution and tissue function. With aging, alterations in fiber organization, cross-linking, hydration, and matrix–cell interactions progressively impair dermal performance, even before structural changes become visible.
INTRODUCTION
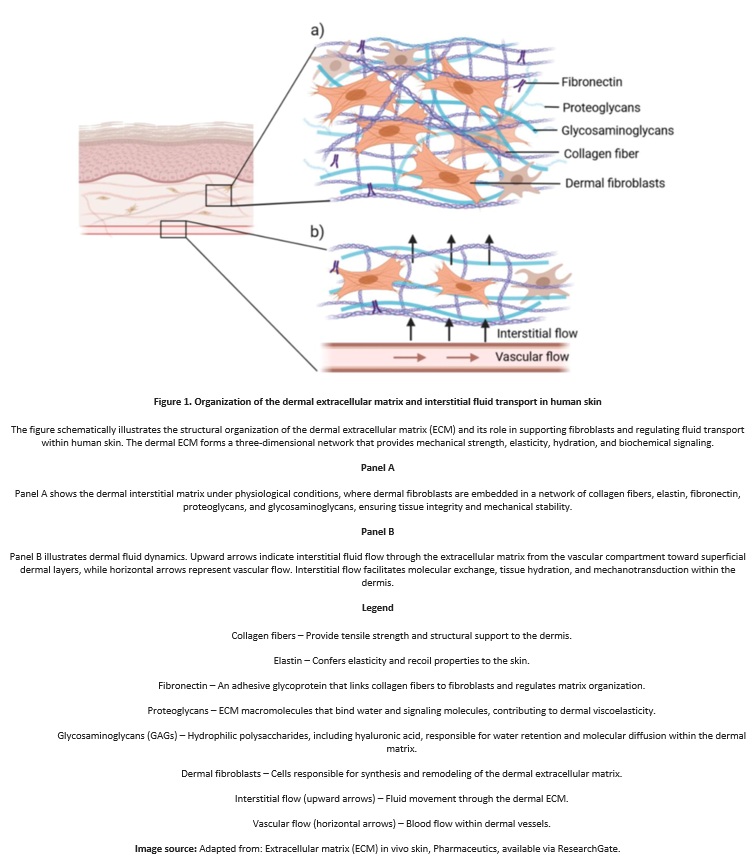
Skin structure and function depend on the coordinated interaction of collagen fibers, elastin fibers, and the extracellular matrix. These elements form a unified network responsible for tensile strength, elastic recoil, mechanotransduction and tissue maintenance.
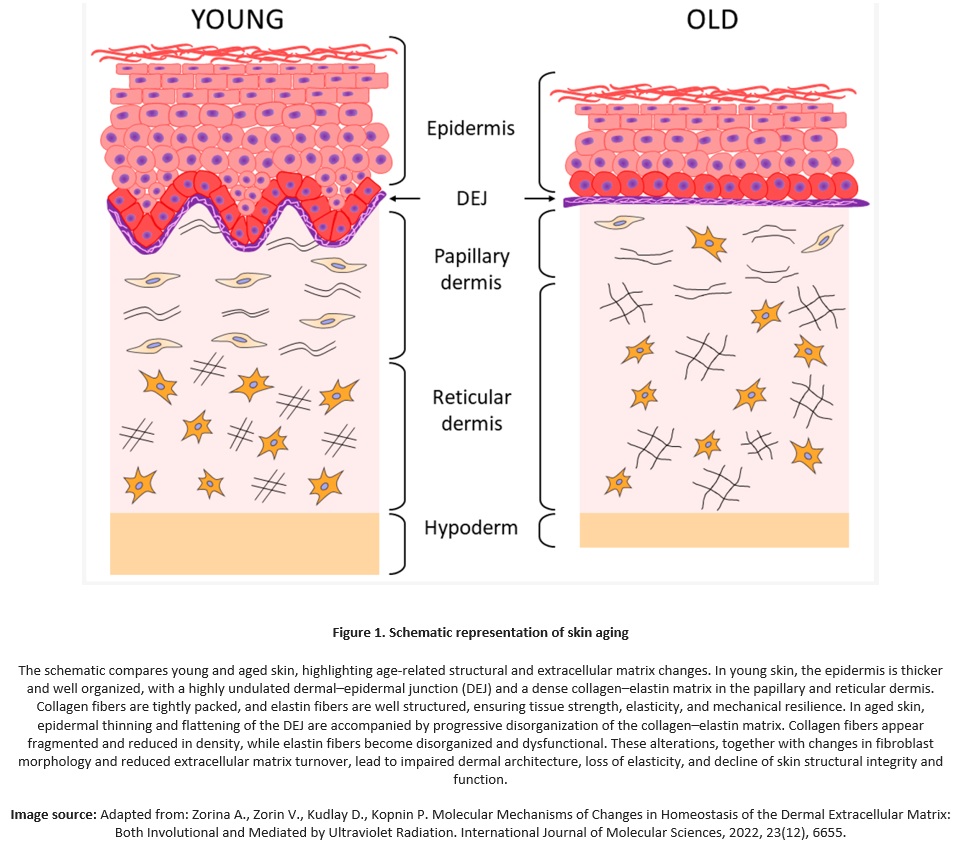
Their organization and interaction determine the skin’s ability to adapt to mechanical stress and support regeneration. With aging, this coordination is progressively disrupted. Changes in fiber architecture, abnormal cross-linking (excessive binding between fibers reducing flexibility), glycation (sugars-related stiffening of proteins), reduced hydration, and impaired matrix–cell signaling decrease mechanical efficiency and regenerative capacity.
COLLAGEN—ELASTIN MATRIX AND EXTRACELLULAR MATRIX IN HUMAN SKIN
Skin structure and mechanical properties depend on the integrated organization of collagen, elastin, and the extracellular matrix (ECM). Collagen and elastin are structural proteins synthesized by dermal fibroblasts and assembled extracellularly into organized fiber systems that gives strength, elasticity, and mechanical stability.
Collagen type I is the predominant fibrillar collagen, accounting for approximately 80–85% of dermal collagen and providing tensile strength and firmness, particularly within the reticular dermis. Collagen type III is closely associated with type I, is more abundant in young skin and during tissue repair and contributes to flexibility and active dermal remodeling. Collagen type V, although present in small amounts, plays a key regulatory role by controlling collagen fibril assembly and diameter. At the dermal–epidermal junction, collagen type IV forms a network within the basement membrane that supports epidermal adhesion and metabolic exchange, while collagen type VII forms anchoring fibrils that ensure stable attachment between epidermis and dermis.
Elastin is assembled into elastic fibers that allow skin stretch and recoil and shows very limited turnover after early adulthood, making elastic fiber damage largely cumulative. Collagen and elastin fibers are embedded within the ECM, a highly hydrated network composed mainly of water, proteoglycans, glycosaminoglycans, and adhesive glycoproteins that regulates hydration, diffusion, and cell–matrix interactions. A well-preserved ECM is essential for proper fiber organization and fibroblast function. When ECM composition or hydration is altered, collagen and elastin lose efficiency and structural organization, contributing to skin ageing even when their overall abundance is relatively maintained.
TURNOVER, AGING, AND DEGRADATION
Collagen renewal occurs through continuous fibroblast activity and takes months to years in adult skin. From the mid-20s, collagen decreases by approximately 1 percent per year on average, with collagen type III declining earlier and faster than type I. With aging, collagen fibers become fragmented, disorganized, and stiffer, reducing mechanical efficiency even before significant quantitative loss. Elastin renewal in adulthood is minimal. Elastic fibers progressively undergo fragmentation and loss of microfibrillar support, leading to reduced recoil and increased stiffness.
Because regeneration is limited, elastin damage accumulates over time. Collagen degradation is driven mainly by matrix metalloproteinases (MMPs), activated by ultraviolet radiation, oxidative stress, and chronic inflammation. Glycation (non-enzymatic binding of sugars to proteins, forming advanced glycation end products, AGEs) creates rigid cross-links that impair collagen flexibility and function. Elastin degradation is driven by elastases, UV exposure, oxidative stress, inflammation, and mechanical fatigue. The extracellular matrix is also affected by aging.
Hyaluronic acid content decreases, hydration is reduced, proteoglycan structure changes, and chronic inflammation accelerates matrix breakdown. ECM degradation further impairs collagen and elastin organization and fibroblast function.
STRUCTURAL AND FUNCTIONAL IMPACT OF MATRIX DEGRADATION
Degradation of collagen (all types), elastin, and the extracellular matrix (ECM) directly alters skin structure and function. Collagen fragmentation reduces dermal thickness, strength, and resistance. Loss of collagen type III limits adaptive remodeling, while degradation of collagen types IV and VII weakens the dermal–epidermal junction, reducing epidermal support. Elastin degradation causes loss of elasticity and recoil, increasing tissue stiffness and reducing the ability to absorb mechanical stress.
Because elastin regeneration is minimal, damage accumulates over time. ECM degradation reduces hydration, nutrient diffusion, and mechanical cushioning. Altered cell–matrix signaling lowers fibroblast activity, slowing collagen synthesis and tissue regeneration. As mechanical support and signaling decline, microvascular efficiency and oxygen diffusion decrease, further limiting cellular metabolism and repair capacity.
This creates a feedback loop in which reduced regeneration accelerates structural deterioration. Together, these changes lead to reduced dermal density, impaired epidermal support, slower regeneration, decreased resilience, and increased vulnerability to external stress. Supporting skin integrity requires addressing its multidimensional nature, which includes maintaining hydration, limiting chronic inflammation and oxidative stress, supporting fibroblast function, improving skin oxygenation and nutrient exchange, and preserving structural organization. Together, these factors sustain mechanical strength, elasticity, and regenerative capacity over time.
SUPPORTING THE COLLAGEN—ELASTIN—ECM NETWORK IN COMPROMISED SKIN
In compromised skin, degradation of the collagen–elastin matrix and the extracellular matrix slows regeneration, alters microcirculation, and sustains low-grade inflammation. Tissue recovery becomes delayed and unstable, requiring controlled, physiology-based, non-invasive support that respects tissue integrity and biological timing. Because collagen and elastin regenerate over weeks to months, limiting inflammatory load is essential to prevent further matrix breakdown.
Restoring microcirculation, renewal, hydration, and structural organization support fibroblast activity and promotes gradual tissue recovery. A multidimensional, non-invasive approach such as JetPeel by TavTech enables this support through a needle-free trans-epidermal delivery system, allowing targeted ingredients, enriched with osmolytes aimed at invigorating collagen and elastin, to be introduced in a stage-specific manner fully aligned with regenerative physiology. The uniqueness lies in the ability to support regeneration without needles, without tissue trauma, and without inducing additional inflammatory load, allowing physiological repair processes to proceed in a controlled and biologically coherent manner.
CONCLUSION
Aging-related skin changes arise from progressive dysfunction of the collagen–elastin–extracellular matrix network rather than isolated loss of collagen or elastin. Altered organization, hydration, and signaling compromise mechanical stability and tissue regeneration. Supporting this system requires non-traumatic, physiology-based approaches that respect biological timing and limit additional inflammatory stress.
JetPeel by TavTech non-invasive trans-epidermal delivery technology, together with targeted JetCare by TavTech formulations selected according to tissue condition and regenerative phase, supports physiological skin regeneration. This approach supports microcirculation, hydration, oxidative balance, and skin structural and function without inducing mechanical or inflammatory damage, contributing to gradual functional recovery and long-term skin rejuvenation.